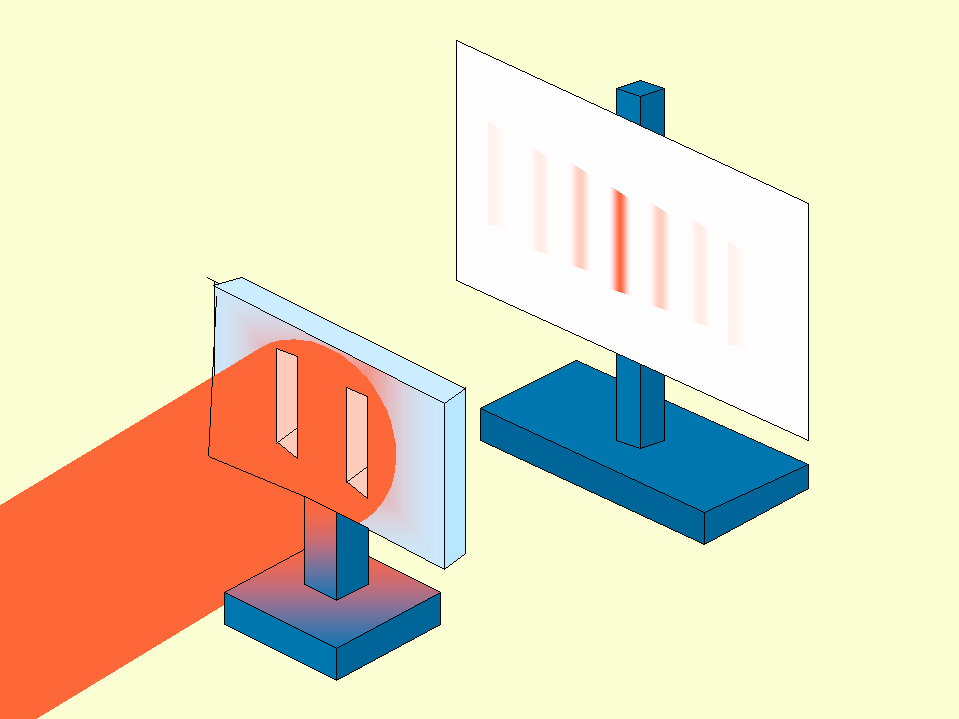
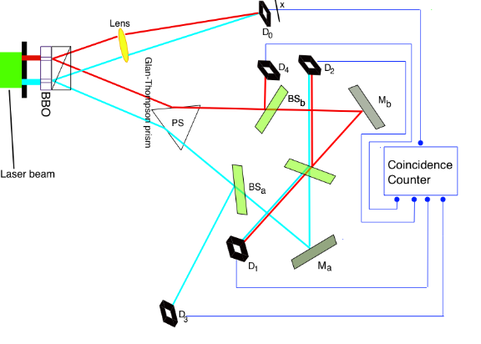
Author- BY NAIDU MAHENDRA.S | 29-JAN-21 Why does the effect always follow the cause? Why can’t an effect precede its cause? The thought itself that an effect might precede its cause appears nonsensical at first. If a table is laid, then it can only be because someone has laid it. Let’s look at an example. If a glass of wine is swung off a table by one’s hand, for anyone who sees the situation, it is obvious that the swinging of hand is the cause and falling of wine glass is the effect. However, why can’t be it visa versa? Why is it not possible for the falling of wine glass to be the cause and swinging of hand to be its effect? Is there any way for a falling wine glass to cause your hand to swing before its own falling? If by any chance the answer is yes then not only our view towards cause and effect will change, but it will also change our perception of seeing past, present and future. Delayed Choice Quantum Eraser Experiment: There is an experiment called Double slit Experiment in which the photon is shot one at a time towards a screen with a barrier having two slits in front of it. In this experiment, if interference pattern appears on the screen then it shows wave nature and if it does not show any interference pattern then it indicates particle nature. Though light is a wave and a single photon is particle yet interference pattern that appeared on the screen shocked the physicist because photon showing wave nature is only possible if a single photon was passing through both the slits simultaneously which was impossible. So, the physicist repeated the experiment. Despite numerous repetitions, the interference pattern showed its absoluteness every time.
The physicist then tried a different approach. They put detectors on each slit to confirm the fact that the photon is simultaneously passing through both slits. Surprisingly, this time, the photon only passed through one of the slits and showed a non-interference pattern which indicated that the photon was a particle. This result was even more shocking because somehow photons knew that they were detected which is why they denied to show interference pattern whenever the experiment was carried out with detectors. So, finally physicists tried a completely different approach with a setup similar to the diagram shown above. They placed a crystal right behind the slits which can produce a pair of entangled particles from each photon, one of which was sent to screen and another was sent to a detector which was at a long distance from slit as compared to the distance between slit and screen. In this case one of the particles have to strike on screen before the other reaches the detector. So, this time there was no way that photon could escape. But this time also the result was non-interference pattern. Though the photon sensed the detector after striking the screen still it showed non-interference pattern which simply means that the moment photon sensed detector it knew that it was too late for sensing a detector and showing non-interference pattern, so it just time travelled to past with the information of detector being present to the moment when it was shot from a source and thus it changed everything. Thus, in this case the cause was the photon sensing the detector in present and effect was its changing pattern by travelling to the past. We can conclude from this experiment that its not always our past which affects the future but instead future can also change our past. Its not always the cause who comes first, it can be an effect too. So, next time before deciding which one is cause and which is its effect, think twice. Who knows the thing which you consider cause might turn out to be an effect?
0 Comments
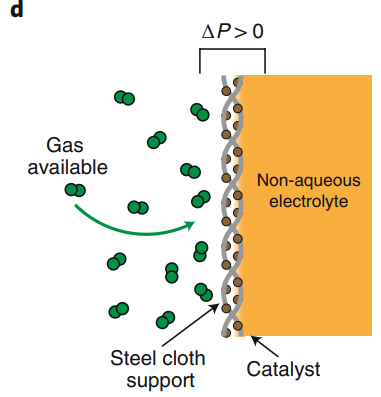
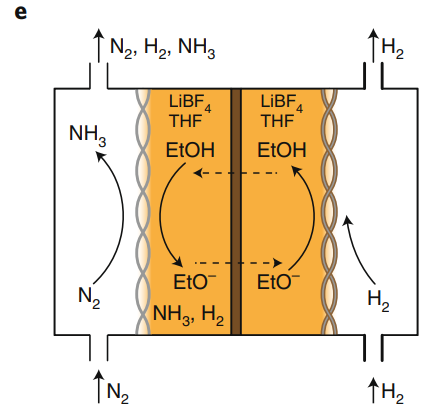
Author- By SUPRAJA G.S | 22 January 2021 A Recent Chemical Engineering finding. Ammonia is a key chemical produced in huge quantities worldwide. it is a major raw material for fertilizer industries. It is also used in the manufacture of Nitric acid, Hydroxylamine, Hydrazine, Amines, and amides, and in many other organic compounds, plastic, fibers, pharmaceuticals, refrigerants in heat pumps, and even explosives all use ammonia as raw material. Moreover, ammonia has been suggested as a hydrogen carrier recently because of its high hydrogen content. Its primary industrial production is via the Haber-Bosch method. A method of directly synthesizing ammonia from hydrogen and nitrogen, developed by the German physical chemist Fritz Haber. In the Haber-Bosch process, which is the main method of ammonia synthesis, nitrogen reacts with hydrogen using a metal catalyst to produce ammonia. However, this industrial process is conducted at 200 atm(atmospheric pressure) and high reaction temperatures of nearly 500°C. Additionally, ammonia production requires using much natural gas, so scientists have been looking for alternative methods for the past two decades to sustainably synthesize ammonia at low temperatures. Several alternatives to the existing process have been proposed, including the electrochemical synthesis. In the recent finding, A rapid and efficient electrochemical reaction requires an active catalytic material, as well as a proper electrode and cell design. Now, a gas diffusion electrode based on a stainless steel cloth successfully overcomes gas transport limitations for high-current ammonia electrosynthesis in non-aqueous solvents at ambient conditions, has been invented. PROCESS: Lithium metal can split the nitrogen triple bond spontaneously at ambient conditions, forming lithium nitride (Li3N), which then reacts with a proton source to form ammonia. However, the lithium-mediated approach requires non-aqueous electrolyte and proper design of the gas diffusion electrode to guarantee high ammonia yields and minimization of diffusion phenomena. A hydrophobic carbon-fiber gas diffusion electrode with an aqueous electrolyte, where a well-defined three-phase boundary exists. The use of a gas diffusion electrode is necessary to enhance the solubility of the reactant gases in the electrolyte and to maximize their diffusive transport to the catalyst surface. However, if a non-aqueous solvent is used, deep penetration of the electrolyte into the carbon fibrous structure occurs, thus flooding the electrode and slowing diffusion transport of the gases, as shown in Fig. The use of stainless steel cloth (SSC) as a gas diffusion electrode for ammonia electrosynthesis to overcome transport limitations of gaseous reactants (H2 at the anode and N2 at the cathode) in non-aqueous solvent (that is, tetrahydrofuran) systems. Electrolyte penetration into the cloth is controlled by applying a small pressure gradient across the gas diffusion electrode. This pressure gradient prevents electrolyte to flow into the gas compartment, and establishes an efficient gas–electrolyte interface, as shown in Fig. The investigation was performed separately at the anode (with Pt deposited on SSC) for the hydrogen oxidation reaction, at the cathode (with Li deposited on SSC) for the reduction of nitrogen to ammonia, and finally, in a coupled electrochemical Haber–Bosch system using both electrodes. Ethanol was used as the proton source at the cathode — to produce ammonia — and is then regenerated from protons coming from the anode.
The original approach proposed by Manthiram and co-workers clearly shows that in ammonia synthesis, and more generally in all electrochemical reactions that are potential candidates to replace high-energy-consuming industrial processes, electrode and cell design are of paramount importance to find new engineering solutions and obtain high efficiencies and performance. This is a call for scientists in the multidisciplinary fields of industrial chemistry, chemical engineering, and catalysis to focus their research on these areas. |